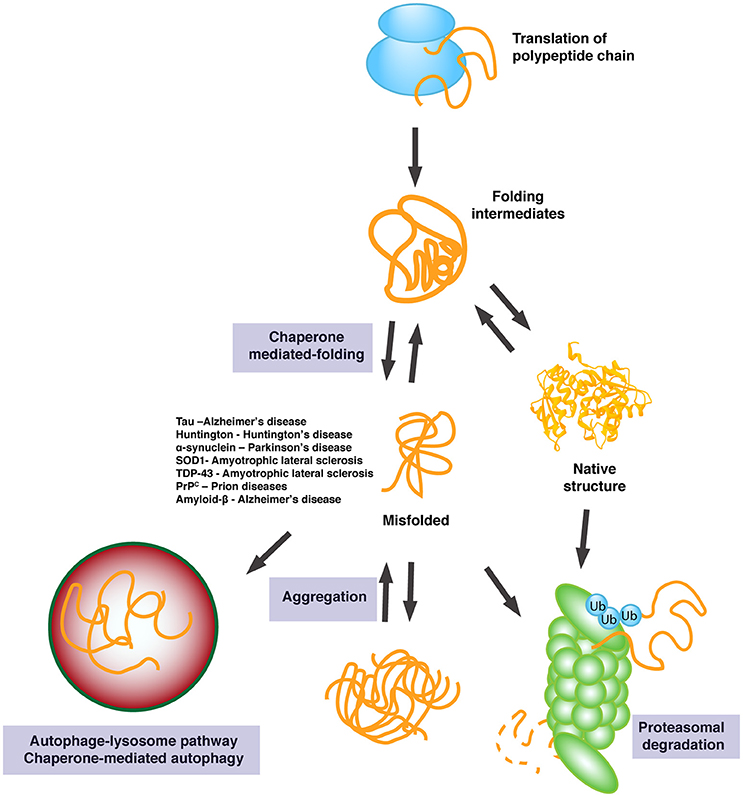
Heck and Doryen Bubeck, is published in Nature Communications. "Structural basis of soluble membrane attack complex packaging for clearance," by Anaïs Menny, Marie V. For example, beta-amyloid fibers, if they are allowed to accumulate, can lead to the plaques that characterize Alzheimer's disease.įirst author Anaïs Menny, also from the Department of Life Sciences at Imperial, said: "If clusterin uses the same method to recognize and prevent beta-amyloid accumulation as it does for MACs, then we could get some really interesting insights into how the early precursors to Alzheimer's disease arise." It's this prevention of build-up of further material that the researchers say could provide interesting insights into other material accumulations. Chaperones mediate the proper folding, stability and/or activation states of their substrate proteins. There are several chaperone systems which carry out a multitude of functions all aimed towards insuring the proper folding of target proteins. It then prevents the MAC from building up more of the components it needs to fully assemble and carry out its hole-punching attack. Chaperones are an essential group of proteins necessary for cell viability under both normal and stress conditions. "Seeing how these proteins stop MAC provides the first clues into how this branch of the immune system can be controlled and shows us how these chaperones might capture other harmful proteins in the blood."įrom their detailed investigation, the team showed that clusterin attaches to a precursor version of MACs that is dissolved in the bloodstream. events are monitored by specialized proteins, chaperones, and proteases. Here we discovered how chaperones in the blood capture rogue molecules and prevent them from damaging human cells. Switch from Chaperone to Protease in a Widely Conserved Heat Shock Protein. Doryen Bubeck, from the Department of Life Sciences at Imperial, said: "When a pathogen is detected, our immune system goes into overdrive to make MACs and not all of them reach their bacterial targets. /SHap-r-rn//pr-tn/: protein complexes that guide correct protein folding, through a variety of methods. Now, the team from Imperial and institutions in the Netherlands have managed to capture and investigate in unprecedented detail MAC precursor molecules bound to the chaperone proteins, revealing how the chaperones stop MACs becoming fully functional. 
Scientists knew special chaperone proteins-called clusterin and vitronectin-helped to prevent these MACs from causing unwanted attacks, but didn't know how. The immune system therefore creates loads of MACs when an invader is detected, but not all of these reach their targets, meaning many end up in the bloodstream, where they could potentially damage the body's own cells. If enough holes are punched, the bacteria will pop and die. The team studied membrane attack complexes (MACs) – components of our immune system that punch minute holes in the membrane of invading bacteria. It could also point to how special chaperone proteins can prevent the accumulation of other harmful molecules, such as those associated with Alzheimer's disease. coli via the Erwinia chrysanthemi Hrp system.The research, led by Imperial College London scientists and published today in Nature Communications, provides insights into how our body keeps the immune system in check. In contrast, WtsF was needed for efficient delivery of WtsE from E. However, WtsF was required for full accumulation of WtsE within the bacteria at low temperatures. stewartii, and the virulence of wtsF mutants was reduced only at low inoculum concentrations. WtsF, the putative chaperone of WtsE, was not required for secretion of WtsE from P. They are highly ubiquitous and assist the folding of newly synthesized proteins as well as the refolding of partially folded proteins into their three-dimensional structures 52, 53, 56. WtsE-induced cell death was not inhibited by cycloheximide treatment, unlike the hypersensitive response caused by a known Avr protein, AvrRxo1. Molecular chaperones are defined as a class of proteins that interact with, stabilize and help proteins to acquire their native conformation 52. Delivery of WtsE into sweet corn leaves by an Escherichia coli strain carrying the hrp cluster of Erwinia chrysanthemi caused water-soaking and necrosis. An intact C-terminus of WtsE, which contained a putative endoplasmic reticulum membrane retention signal, was important for function of WtsE. WtsE, a member of the AvrE family of effector proteins, was essential for pathogenesis on corn and was complemented by DspA/E from Erwinia amylovora. In this study, we genetically and functionally characterized a disease-specific (Dsp) effector locus, composed of wtsE and wtsF, that is adjacent to the hrp gene cluster. stewartii to sweet corn and maize requires a Hrp type III secretion system. The pathogenicity of Pantoea stewartii subsp.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
